





The Irish dairy sector exports 4.5 million tonnes of product with a value of 2.2 billion per annum2. Ireland’s climate and soil favour the life cycle of the liver fluke parasite (Fasciola hepatica) and its intermediate host the mud snail (Galba truncatula). Consequently Irish livestock are subject to some of the highest liver fluke infection levels in the world.

Liver fluke has 3 stages – early immature, immature and adult – all of which cause liver damage. The earlier you kill liver fluke in dairy cows the better.
By killing early immature liver fluke, FASINEX 240 stops further liver damage caused by the immature and adult stage liver fluke from ever occurring.Liver condemnations at slaughter or acute liver fluke disease are obvious losses. However, subclinical disease is far more important resulting in reduced:

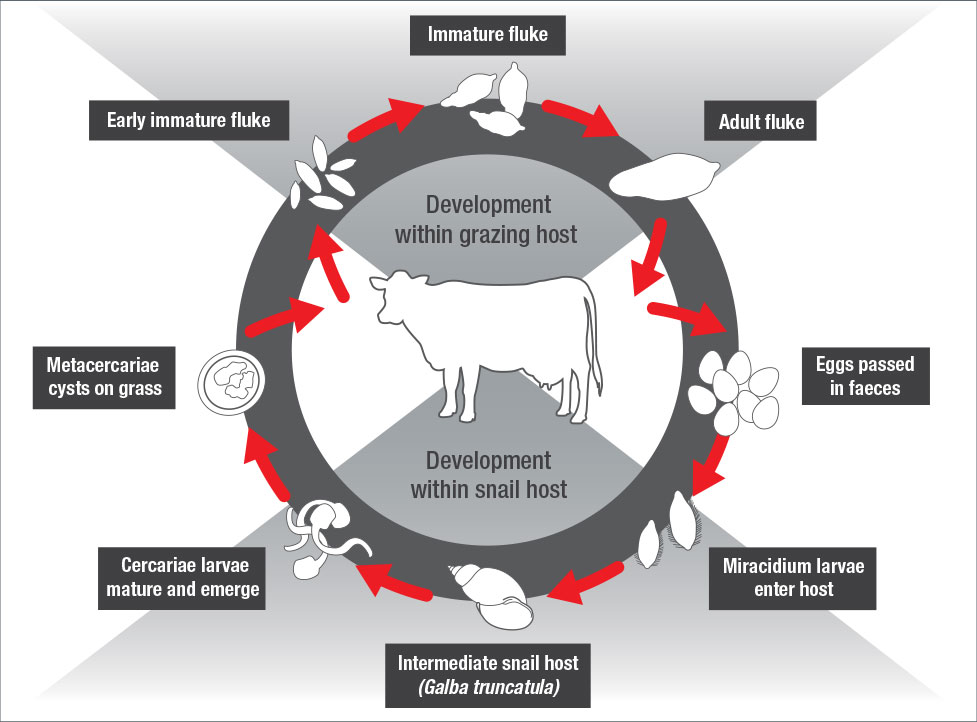
Liver Fluke has a complex lifecycle with a main host (Cattle and sheep) and an intermediate host (the mud snail).

To be able to treat and manage this complex problem effectively – and limit its detrimental effects on dairy cow performance, health and welfare – it is vital to know the status of your herd.
Some products leave the early immature and immature stages alive. Only triclabendazole, in FASINEX 240, kills all 3 stages of liver fluke, reducing liver damage and pasture contamination.
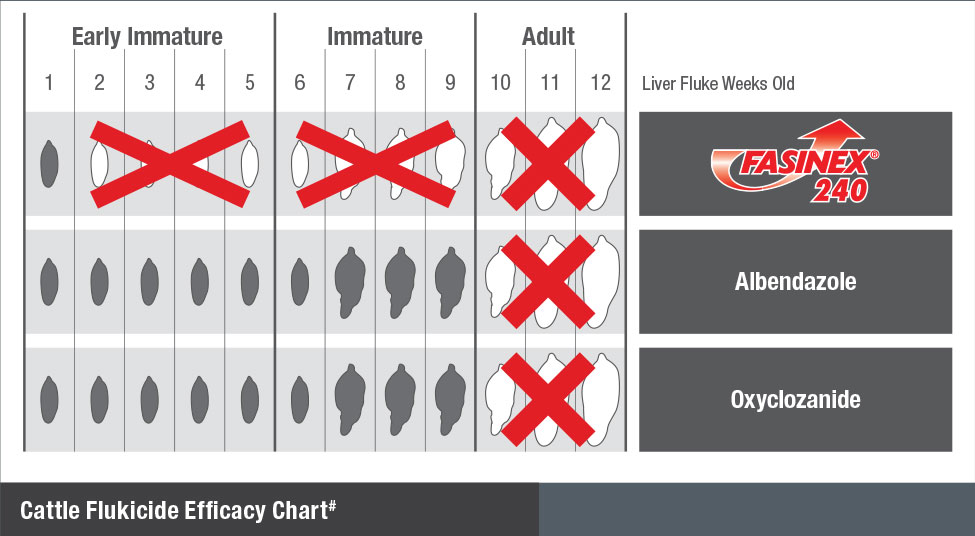
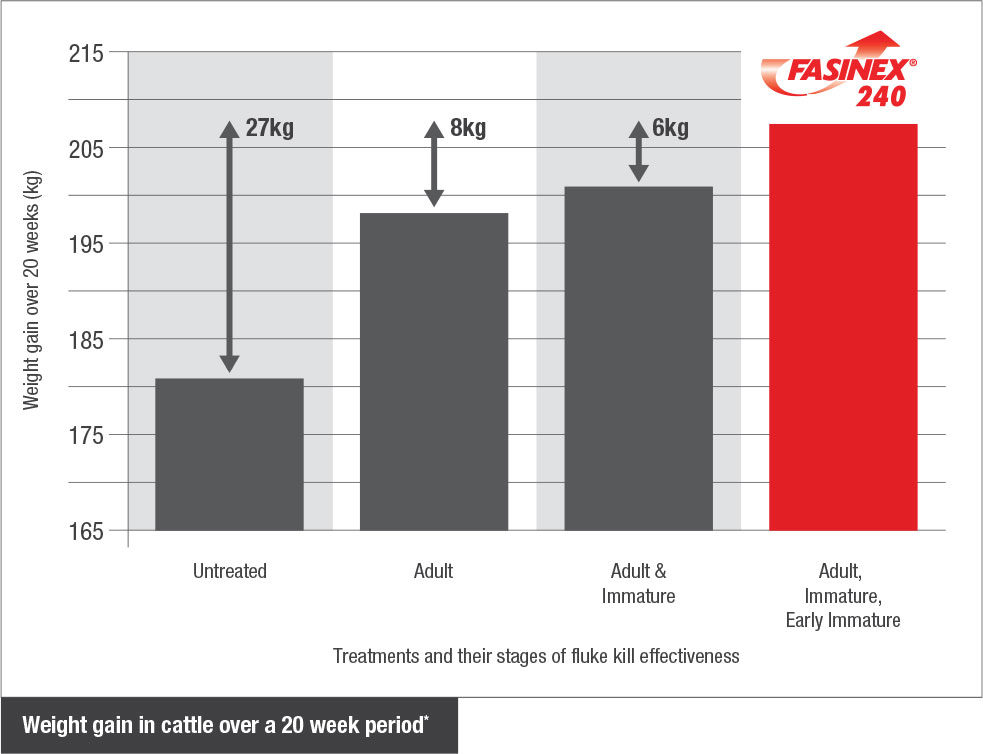
Killing all 3 stages of liver fluke means less liver damage, less blood loss, better growth and productivity.
A study comparing live weight gain of animals treated for all 3 stages of liver fluke, compared to animals treated for adult liver fluke only, demonstrated a benefit of 8 kg over a 20 week period9.
Because it kills liver fluke at earlier development stages, Fasinex 240 achieves the same level of liver fluke kill with fewer doses than oxyclozanide or albendazole. This could save you time and money, improving productivity and reducing the need to handle cattle to eliminate the remaining liver flukes.
For the treatment of acute, subacute and chronic infection due to early immature, immature, and mature stages of Fasciola hepatica. If infected animals are treated before disease has developed, fasciolosis can be prevented.
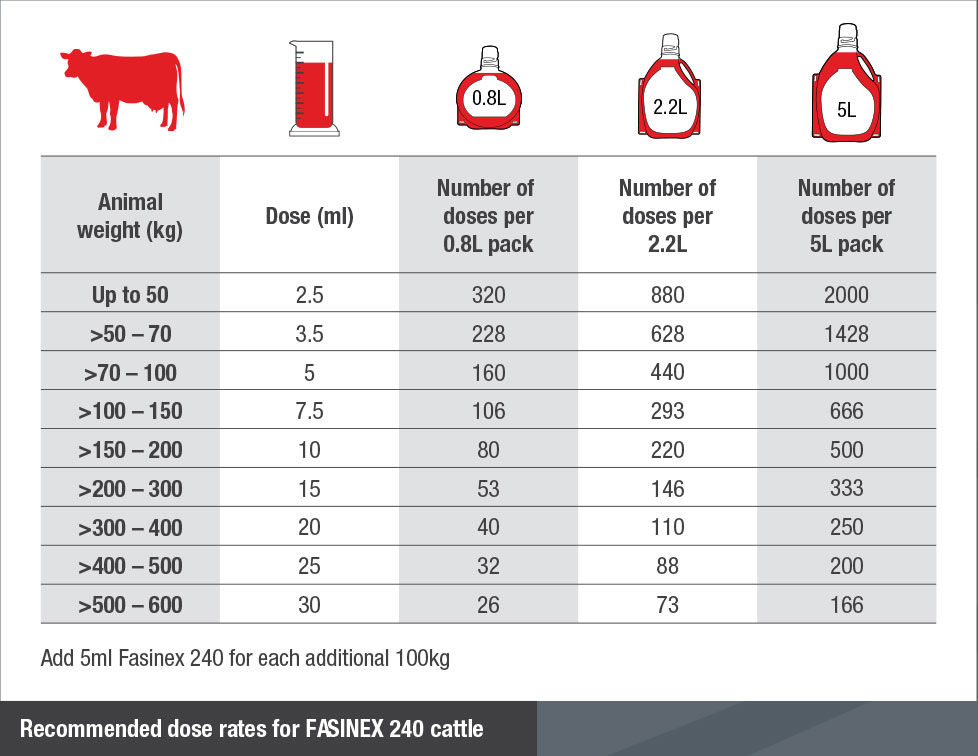
To ensure administration of a correct dose, bodyweight should be determined as accurately as possible; accuracy of the dosing device should be checked. If animals are to be treated collectively rather than individually they should be grouped according to their bodyweight and dosed accordingly, in order to avoid under- or over-dosing. Administer 5 ml/100 kg bodyweight, equivalent to 12 mg triclabendazole per kg of bodyweight. FASINEX is administered orally after thorough shaking of the suspension. Most types of automatic drenching guns are suitable.
Clean drenching gun before and after use. FASINEX can safely be given to young, pregnant or stressed cattle. However, the product is not permitted for use in lactating animals producing milk for human consumption. Fasinex 240 is given once. The administration may be repeated several weeks or months after the first treatment depending on the epidemiological situation. In case of acute fasciolosis, treat immediately, and repeat on the advice of the prescriber, depending on the ongoing level of challenge.
On land where sheep are being treated according to the preventative programme and where cattle are also grazing these areas, Fasinex 240 should be administered to the cattle on the same treatment dates as the sheep. Treatment times should be customised under veterinary advice for each individual farm. Spring/summer treatments prevent the liver flukes entering the mud snail and so the life cycle is broken. Every animal on the same land including sheep should be included in the programme and all animals should be treated on the same day. Fasinex 5% should be used in sheep. All bought in animals should be dosed before joining the main herd.
Areas of Average Liver fluke Infection:Dose all cattle exposed to liver fluke infected pastures at regular intervals of 8 -10 weeks throughout the liver fluke season.
Housed cattle:Dose cattle, which have grazed liver fluke infected pasture in the Autumn and at the time of or shortly after housing. Dosing should start at the beginning of the liver fluke season when animals are still outdoors.
Bought in cattle:Dose before joining the main herd.
Treatment of acute outbreaks:The herd should be treated immediately after diagnosis and veterinary advice should be sought for subsequent dosing intervals.
Do not eat, drink, or smoke while handling the product. Wash hands and exposed skin after handling the product. Wash splashes from eyes and skin immediately with water. Remove contaminated clothing. Clean drenching equipment before and after use.
Keep out of the reach and sight of children.
FASINEX is safe for use in all stages of pregnancy and lactation. However, the product is not permitted for use in lactating animals producing milk for human consumption.
Not intended for use within 35 days of calving. Do not use in animals with a known hypersensitivity to the active ingredient. Do not administer Fasinex 240 to sheep.
Care should be taken to avoid the following practices because they increase the risk of development of resistance and could ultimately result in ineffective therapy.
Suspected clinical cases of resistance to anthelmintics should be further investigated using appropriate tests (e.g. Faecal Egg Count Reduction Test). Where the results of the test(s) strongly suggest resistance to a particular anthelmintic, an anthelmintic belonging to another pharmacological class and having a different mode of action should be used.
Any unused veterinary medicinal product or waste material derived from such veterinary medicinal products should be disposed of in accordance with national requirements. Do not contaminate ponds, waterways or ditches with the product or used container.
Shake the container before use. Once opened, use within 12 months. Store in tightly closed original container.
For further information about Fasinex 240 contact:
Elanco Animal Health, Lilly House, Priestley Road, Basingstoke, Hampshire, RG24 9NL.
Tel +44 (0)1256 353131 or in Ireland Tel 051 377201.
Fasinex® 240 contains 24% w/v triclabendazole LM.
Fasinex® 240 is a registered trade mark owned by or licensed
to Eli Lilly and Company, its affiliates or subsidiaries. Advice on
the use of this or alternative medicines must be sought from the
medicine prescriber.
Licensed Merchant. IEDRYFAS00001 rdp19903/06/15
Use medicines responsibly
1. Oakley G.A, et al 1979, Veterinary Record, 104, 503 - 507.
2. Moorepark dairy levy research update, 4/5/2011, Kilkenny Greenfield dairy programme.
3. Irish Veterinary Journal Volume 63, Number 11, pg 702.
4. DeLiver project www.deliver-project.eu.
5. University of Reading, Department of Agriculture
and Food Economics, The Economics of Fascioliasis (Liver Fluke).
6.Department of Agriculture and Rural Development, Technical Note, Dairy,
13th April 2003.
7. M.C Lopez – Diaz et al, Theriogenology, 1998.
8. Boray J.C, Chemotherapy of fascioliasis (Research done by Ciba-Geigy Inc).
9.Boray, JC, NSW, Veterinary Proceedings, Vol 18, 1982.
* Boray JC, NSW Veterinary Proceedings, Vol 18, 1982.
# Claims based on published summary of product characteristics for
products containing the active ingredients specified. Information accurate
on the date of publication of this detailer, September 2012.